Help Wanted – NASA
As NASA continues to prepare to resume human spaceflight launches and readies its ambitious ‘Journey to Mars’, it is putting out its “help wanted” sign.
The space agency announced that it will soon begin to accept applications for its next class of astronauts who will fly from the Space Coast of Florida aboard new American-made commercial spacecraft.
Qualifications required to become a NASA Astronaut candidate, include U.S. citizenship, at least a bachelor’s degree, although an advanced degree is preferred, in either engineering, mathematics, or biological or physical science from an accredited institution.
The space agency said that candidates must also have at least three years of related and increasingly responsible professional experience or at least 1,000 hours of command pilot time in jet aircraft and be able to pass NASA’s long-duration spaceflight physical.
Applications to become an Astronaut candidate will be accepted by the space agency from December 14th until the middle of this coming February.
NASA will take about a year and a half to go through all the applications and will announce the new astronaut candidates sometime in mid-2017.

Engineers from Onsala Space Observatory’s Group for Advanced Receiver Development examine the top part of SEPIA before its installation at APEX in Chile. (ESO/Sascha Krause)
New Device Helps Find Water in Space
Astronomers have a new tool that will help them look for water throughout the universe.
The Swedish-ESO PI receiver for APEX or SEPIA can detect light signals in the 1.4 to 1.8 millimeter wavelength range, where astronomers say water in space can be found.
The SEPIA device was installed on the European Southern Observatory’s Atacama Pathfinder Experiment, or APEX, telescope located on the incredibly dry Chajnantor Plateau some 5000 meters above sea level in the Chilean Andes.
Atmospheric water vapor at most places on Earth would hinder the space water search effort, but the exceptional dry conditions at their Chilean site, according to ESO astronomers, makes it an ideal location to look for celestial water.
Scientists say that water is important in a number of astrophysical processes, such as star formation, and that it may also play a key role in the origin of life.
Devices identical to SEPIA are also being installed in the antennas of ESO’s Atacama Large Millimeter radio telescope array.
Diamonds May Not Be So Rare After All
A new study from Johns Hopkins University suggests that diamonds may not be as rare as previously believed, and that formation of the carbon-based gem may be a much more common process than thought.
But before you get excited about picking up a big diamond ring at a cheap price, the researchers add that the diamond bonanza is actually in the deep Earth. The very deep earth, actually, say the researchers behind the study.
To get to any of the diamonds being formed deep below our feet, they must first be brought to near the Earth’s surface by somewhat rare volcanic magma eruptions. The researchers also add that most of these diamonds aren’t the large and gem quality stones found in valuable jewelry, but are so tiny that you’d need a microscope to seem them.
The researchers also found that diamonds could be formed by a natural chemical reaction that is much easier than the commonly understood process involving high temperatures and tremendous pressure.
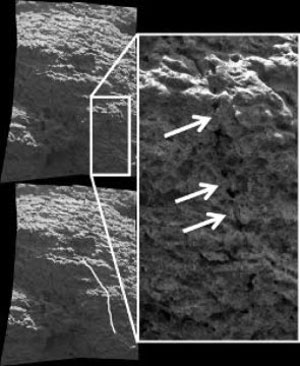
This Microscopic Image Mosaic shows a pre-existing crack which is being “healed over”, which is evidence for the gel weathering alteration process. (NASA/JPL & S. Cole)
Ancient Martian Rock Attacked by Acidic Fog
Planetary scientist Shoshanna Cole from Ithaca College in New York says that she has found evidence that that some type of acid fog created by past volcanic activity dissolved rocks on Mars.
Acid fog, or VOG as it’s known on Earth, is created when volcanic gas reacts with oxygen and sunlight.
Blankets of VOG form from time to time near Hawaii’s Kīlauea volcano, which has been continuously erupting since 1983.
Cole used a variety of data gathered in the Husband Hills area of Mars’ Guseve Crater by NASA’s Spirit rover to make her findings.
The data allowed her to hypothesize that the rocks in this area of Mars were exposed to acidic water vapor from volcanic eruptions over a long period of time.
She says that after the Martian VOG settled on the rocks it dissolved some of its minerals and formed a gel. Later, after the gel’s water evaporated it left behind material that broke down the rocks.
Uninterrupted Sleep Helps You Wake Up in a Good Mood
If you want to wake up and stay in a good mood all day, a new study from researchers at Johns Hopkins School of Medicine suggests that you need to get a good night’s sleep: not a necessarily a lot of sleep, but some good quality uninterrupted sleep.
The researchers found that being roused from sleep several times a night puts a damper on a good mood more than having a small amount of sleep without any interruptions.
According to the study, having continued nights of interrupted sleep can have increasing negative effects on a person’s mood.
Patrick Finan, the study’s lead author and an assistant professor of psychiatry and behavioral sciences at Johns Hopkins, says that when your sleep is disrupted throughout the night, you don’t have the chance to go through each of the various phases of sleep.
Not progressing through the stages of sleep can deprive you of the needed slow-wave sleep necessary to making a person feel refreshed and restored.
























You lost an important information from NASA: the fly to Mars is one way.
What can a person like me do to get access to NASA?
@Jeannine ThompsonThat sounds awful.
Considering days gone by it is clear that the components which were employed,
were those easily available such as seashells,
twigs, and natural gems to mention a couple
of.